What mTOR Signaling Is and Why Longevity Researchers Keep Talking About It
Inside the molecular pathway that controls how your cells grow, clean house, and age, and why scientists believe modulating it could be the most important lever in the future of human healthspan.
mTOR signalling sits at the centre of ageing science, linking caloric restriction, rapamycin, autophagy, and healthspan extension. Here is what the research actually shows, and what it means for you.
There is a moment in every serious conversation about ageing science when someone says the word “mTOR,” and the room either lights up or goes blank.
Trending Now!!:
I have been in both kinds of rooms, including research seminars where the debate over rapamycin dosing turned almost personal, and patient consultations where the person across from me was already sourcing the drug online before we had finished talking.
After more than a decade working at the intersection of cellular biology, metabolic medicine, and longevity research, I can tell you this: mTOR is not just another acronym in a long list of molecular pathways. It is, by many measures, the closest thing we have found to a master dial for biological ageing.
Getting it right matters more than most people realize, because getting it wrong, either by ignoring it entirely or by trying to suppress it too aggressively, carries real consequences for your health.
What mTOR Actually Is
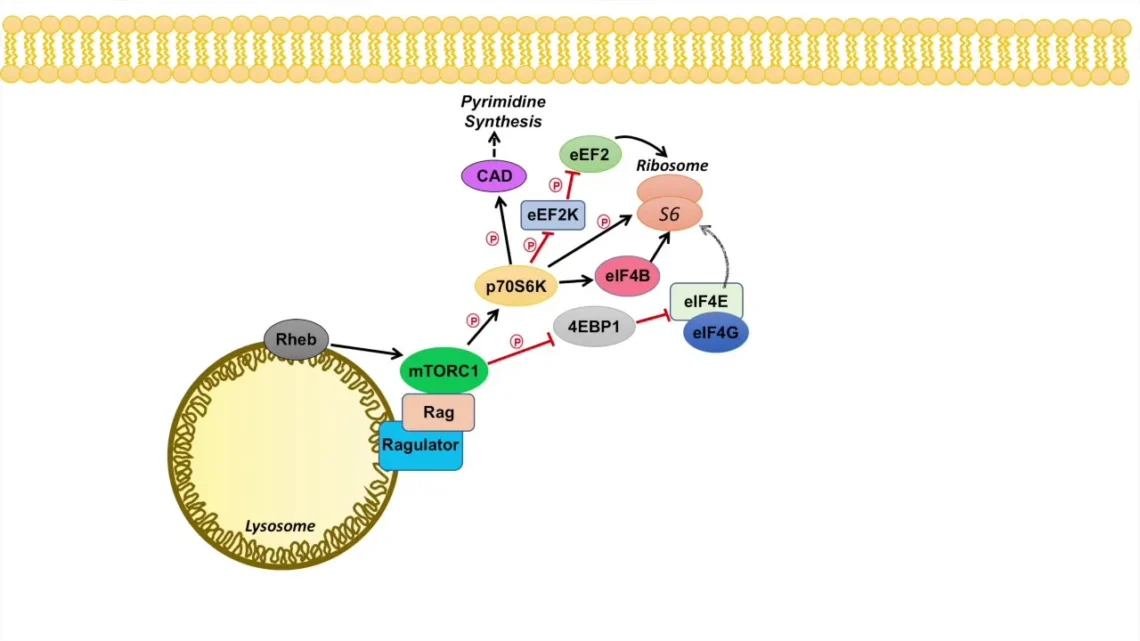
The mechanistic target of rapamycin, or mTOR, is a protein kinase, essentially an enzyme that switches other proteins on or off by attaching phosphate groups to them. It sits inside almost every cell in your body, and its job is to read the environment and decide whether the cell should be growing, dividing, and building things, or slowing down, cleaning house, and conserving resources.
Think of it as a cellular air traffic controller. When nutrients are abundant, growth factors are circulating, and energy is plentiful, mTOR says: build, multiply, go. When the cell is stressed, starved, or damaged, mTOR pulls back and lets the maintenance crews do their work.
mTOR acts as a master regulator of intracellular metabolism, responsible, at least in part, for the longevous phenotype in species where its activity is reduced. That sentence sounds dry in a paper. In practice, it means that how active this pathway is across the decades of your life may have more to do with how well you age than almost anything else researchers have studied.
The Two Complexes: mTORC1 and mTORC2
mTOR does not operate alone. It organizes itself into two distinct protein complexes, each with different partners, different jobs, and different sensitivities to intervention.
mTOR is found in two protein complexes, mTORC1 and mTORC2, that have distinct components and substrates and are both inhibited by rapamycin, a macrolide drug that robustly extends lifespan in multiple species, including worms and mice.
mTORC1 is the better-understood of the two. It is the growth-promoting, autophagy-suppressing complex that most longevity research focuses on. When mTORC1 is highly active, cells are in full production mode: synthesizing proteins, building lipids, and suppressing the recycling process called autophagy. This is healthy and necessary when you are young and rebuilding from exercise or recovering from illness. It becomes a problem when it never turns off.
mTORC2 is less studied but increasingly important. Loss of hypothalamic mTORC2 signalling in mice decreases activity level, increases the set point for adiposity, and renders the animals susceptible to diet-induced obesity.
Mice lacking this pathway displayed higher fat mass, impaired glucose homeostasis throughout life, became more frail with age, and had decreased overall survival. The lesson, which took years to fully appreciate, is that targeting mTOR for longevity is not simply a matter of suppressing everything. The two complexes play complementary roles, and blunt suppression carries costs.
Why Longevity Researchers Are Obsessed With This Pathway
The short answer is that every time scientists have found a way to reduce mTOR signalling in an animal, that animal has tended to live longer. The consistency across wildly different species is what gives researchers pause and excitement in equal measure.
Reduced mechanistic target of rapamycin signalling extends lifespan in yeast, nematodes, fruit flies and mice, highlighting a physiological pathway that could modulate ageing in evolutionarily divergent organisms. When the same result shows up in organisms separated by hundreds of millions of years of evolution, the scientific community pays attention.
The data from flies is particularly striking. Reduction of mTOR in late life results in instant benefits on life expectancy, based on a large-scale demography comprising over 10,000 individual flies. That phrase “instant benefits” carries weight. It suggests that this pathway is not just setting the pace of ageing during development; it is actively influencing survival probability even in animals that are already old.
The German Longevity Study: A Human Signal
For a long time, critics of mTOR research pointed out the obvious gap: the animal data was compelling, but what about humans? A study published in early 2025 in the journal GeroScience began to close that gap in a meaningful way.
Researchers investigated the contribution of rare coding variants to human longevity by analyzing whole exome sequencing data from 1,245 German long-lived individuals and 4,105 geographically matched younger controls.
They identified novel exome-wide significant associations with a significant over-representation of genes involved in mechanistic target of rapamycin signalling. Three rare single variants in the mTOR-pathway genes RPS6, FLCN, and SIK3 were enriched in long-lived individuals.
In plain terms, people who live exceptionally long lives in Germany appear to carry rare genetic variants that naturally tune down specific components of the mTOR signalling pathway. They are not walking around taking rapamycin. Their DNA is doing a version of what the drug tries to do pharmacologically.
The mTOR pathway is involved in modulating growth, sometimes at the expense of longevity. Long-lived individuals may have genetic mutations in the mTOR pathway that optimize for both growth and longevity. That balance, between enough mTOR activity to maintain muscle and tissue, and enough restraint to allow cellular repair, is where the interesting science lives.
mTOR, Autophagy, and the Cellular Cleaning Problem
One of the most important things mTOR does is suppress autophagy. This matters enormously for aging, and it is worth understanding why.
Autophagy, from the Greek for “self-eating,” is the process by which cells break down and recycle their own damaged or dysfunctional components. Misfolded proteins, worn-out mitochondria, cellular debris accumulated over the years: autophagy clears all of it. Without regular autophagy, cells accumulate junk. That junk drives inflammation, mitochondrial dysfunction, and the kind of proteostasis collapse that we associate with neurodegenerative diseases.
mTORC1 normally inhibits autophagy by phosphorylating components of the ULK1 complex, and its inhibition by rapamycin removes this suppression and initiates autophagosome formation.
What this means practically is that when mTOR is chronically elevated, as it tends to be in people eating high-calorie diets, leading sedentary lives, or simply getting older, autophagy gets chronically suppressed. The cleaning crews stop coming. The cellular environment deteriorates. Ageing accelerates.
This is not a theoretical concern. Autophagy sits at the centre of the ageing transition, helping clear damaged proteins and organelles, reduce inflammatory signalling, and maintain mitochondrial function, all processes that decline with age and are disrupted in neurodegenerative and metabolic disease.
The Rapamycin Conversation: Promise, Caution, and the Gap Between Mice and People
No discussion of mTOR and longevity medicine is complete without talking about rapamycin. Originally discovered in soil bacteria from Easter Island in the 1970s, rapamycin became a transplant immunosuppressant before geroscientists recognized its anti-ageing potential. The excitement is real. So is the complexity.
In one short-lived mutant strain of mice, the mTOR inhibitor rapamycin extends maximum lifespan nearly three-fold. Rapamycin also prolongs life in normal mice as well as in yeast, worms and flies, and it prevents age-related conditions in rodents, dogs, nonhuman primates and humans.
The human picture is considerably more complicated. A 2025 review found no clear clinical evidence that the benefits observed in animals apply directly to humans, pointing to the urgent need for larger, better-designed human trials before recommending rapamycin for off-label use to prevent ageing.
That said, some human signals are encouraging. At low doses, mTOR inhibition has been shown to improve immunity in older people, attenuating immunosenescence. Low-dose everolimus improved B and T cell responses to influenza vaccination in older adults, and a Phase 2b clinical trial of RTB101 significantly reduced respiratory tract infections in 652 older participants.
The PEARL Trial and What It Tells Us
The recently published PEARL trial demonstrated that low-dose intermittent rapamycin was well tolerated over one year and resulted in modest changes in biomarkers of biological ageing, though long-term clinical benefits remain to be established.
“Well tolerated” and “modest changes” are not the headline the longevity community hoped for, but they are honest data. The trial also reinforces something that came up repeatedly in my conversations with researchers working in this space: timing and dosing appear to matter as much as the compound itself. Intermittent late-life administration of rapamycin has been shown to extend lifespan in both sexes, underscoring the importance of timing and dosing strategy.
The biohacking community has outpaced the clinical research on this, with an increasing number of adults taking rapamycin off-label, sourced from online pharmacies, often without medical supervision. An increasing number of physically active adults are taking the mTOR inhibitor rapamycin off-label with the goal of extending healthspan, despite concerns over adverse side effects, including immunosuppression and metabolic disruptions. I have spoken with some of these people.
The ones who are thoughtful about it worry about exactly the right things: wound healing, infection susceptibility, and the question of what happens when you suppress mTOR in tissues that still need it.
Caloric Restriction, Intermittent Fasting, and the Dietary Route to mTOR Modulation
Not everyone wants to take a drug to influence their mTOR signalling, and the good news is that you do not have to. The most consistently reproducible, safest, and most accessible ways to modulate this pathway are dietary.
Caloric restriction and intermittent fasting consistently activate AMPK and sirtuins, inhibit mTOR signalling, and enhance autophagy, aligning with improvements in insulin sensitivity, lipid profile, low-grade inflammation, and selected epigenetic ageing measures in humans.
The pathway works like this: when you eat less, or compress your eating into a shorter window, glucose and amino acid levels drop. Lower amino acid levels, particularly leucine, directly reduce mTORC1 activity. Simultaneously, the energy-sensing enzyme AMPK becomes more active, and it directly inhibits mTOR. The cell shifts from growth mode into maintenance mode. Autophagy ramps up. Cellular debris gets cleared. Inflammation goes down.
Under dietary restriction conditions, reduced mTORC1 activity alleviates inhibition of autophagy for clearing damaged cellular components, and mTORC1 inhibition also activates TFEB, a transcription factor that boosts lysosomal biogenesis.
Fasting-Mimicking Diets and the Human Evidence
Fasting-mimicking diets are hypothesized to function as autophagy-engaging interventions, delivering a temporary metabolic signal that shifts cells from growth and storage toward maintenance and repair. The goal is not sustained stress, but a carefully timed trigger that prompts the cell’s cleanup machinery to recycle cellular waste for energy.
This framing changed how I think about dietary interventions in practice. The old model, eat less chronically, is physiologically effective but socially and psychologically unsustainable for most people. The newer model delivers a periodic signal that activates mTOR suppression and autophagy without chronic deprivation, is both mechanistically sound and practically achievable.
Protein Intake: The Uncomfortable Trade-Off
Here is something the longevity influencer space tends to gloss over: protein intake is one of the most potent activators of mTOR, particularly via leucine, the branched-chain amino acid that directly stimulates mTORC1. The high-protein dietary advice that dominates fitness culture, eat 1.6 to 2.2 grams per kilogram of body weight daily to build muscle, is in direct tension with the low-mTOR signalling that appears to favour longevity.
This is not a hypothetical conflict. It is a genuine biological trade-off that researchers are still working through. My own clinical reading of the evidence suggests that the answer is probably time-restricted protein, not protein avoidance.
Consuming adequate protein in a defined window, rather than grazing on it across 16 hours, may allow for both the anabolic benefits and the periodic mTOR suppression that supports cellular health. The data is not conclusive yet, but the mechanistic logic is sound.
mTOR and Age-Related Disease: The Stakes Beyond Lifespan
Longevity research sometimes suffers from a perception problem. People assume it is only about living longer, which sounds like vanity to some and an unsolvable problem to others. But mTOR research is increasingly relevant to the diseases that fill hospital wards right now.
The development of diabetes, cancer, and ageing has all been related to aberrant mTOR signalling. These are not niche conditions. They are the leading drivers of morbidity and mortality in every ageing population on earth.
In cancer biology, mTOR is frequently hyperactivated in tumour cells, which use it to sustain the uncontrolled growth that defines malignancy. In metabolic disease, chronic mTOR overactivation through dietary excess drives insulin resistance by impairing normal insulin signalling feedback loops.
In neurodegeneration, the failure of autophagy that follows from elevated mTOR activity allows misfolded protein aggregates like those seen in Alzheimer’s and Parkinson’s to accumulate unchallenged.
Specific inhibition of mTORC1 to treat ageing-related conditions has become the goal of basic and translational scientists, clinicians and biotechnology companies, who are exploring whether existing mTOR inhibitors can safely prevent, delay or treat multiple diseases of ageing.
The framing has shifted in recent years. Researchers are no longer asking only whether we can extend maximum lifespan. They are asking whether we can compress morbidity, keeping people out of cognitive and physical decline for longer, by maintaining the kind of mTOR balance that the genetic data from long-lived humans suggests is optimal.
The Evolutionary Perspective
One of the things that gives me genuine confidence in this research, beyond the clinical excitement around rapamycin, is the evolutionary story. The mTOR pathway is extraordinarily conserved across species. The mTOR pathway is an evolutionarily conserved metabolic signalling cascade that regulates cell division, growth, and survival.
When you find the same molecular switch operating in yeast, worms, flies, and mammals, and when reducing its activity extends life across all of them, you are not looking at a quirk of a single organism’s biology. You are looking at something fundamental.
The mammalian and mechanistic target of rapamycin signalling pathway is shown as a central regulator of lifespan and ageing, though the underlying influence of the mTOR pathway on the evolution of lifespan in mammals is not yet fully understood.
That gap in understanding is where some of the most interesting current research is happening, particularly in comparative biology, where scientists are studying why some mammals live dramatically longer than others of similar body size, and what their mTOR activity profiles look like.
What This Means for Anyone Who Is Not a Researcher
Most people reading about mTOR will not be running clinical trials or sourcing pharmaceutical-grade mTOR inhibitors. For them, the practical implications of this research are more modest but genuinely meaningful.
The dietary interventions with the strongest mechanistic basis for mTOR modulation, caloric restriction, intermittent fasting, and fasting-mimicking diets, are legal, accessible, and backed by a growing body of human data. Caloric restriction, intermittent fasting, and fasting-mimicking diets activate autophagy and mitochondrial efficiency in ways linked to lower cardiometabolic risk and slower biological ageing.
Resistance exercise, despite activating mTOR acutely, may also support a healthier long-term mTOR profile by improving insulin sensitivity, reducing baseline inflammation, and creating the kind of rhythmic activation-and-rest cycle that appears more beneficial than chronic elevation.
The key phrase, one I find myself using a lot in clinical conversations, is metabolic rhythm. The body does not need mTOR to be permanently high or permanently low. It needs to cycle, responding to real physiological signals rather than being overwhelmed by a constant flood of calories and amino acids.
Overdoing longevity interventions, whether through supplements, fasting, or pharmaceutical tools, can sometimes cause more harm than good. A growing body of research supports cyclical approaches to mTOR suppression and autophagy stimulation, mimicking nature’s rhythms to support long-term metabolic and mitochondrial health.
That is not a call for extreme biohacking. It is a case for eating in reasonable windows, not snacking constantly, getting adequate but not excessive protein, moving regularly, and treating the occasional multi-day dietary restriction with the seriousness it deserves. These are interventions with a genuine molecular basis, not wellness mythology.
Where the Research Is Going
The field is moving fast. Understanding the effects of rapamycin on mTOR and autophagy could enable the development of effective interventions to support human longevity and metabolic health, making mTOR inhibitors a promising direction for further research.
The next generation of mTOR inhibitors is designed to be more selective, targeting mTORC1 specifically while sparing mTORC2, in an effort to capture the longevity benefits without the metabolic side effects that have complicated rapamycin’s clinical use.
AI-driven drug discovery is accelerating this. The mechanistic target of rapamycin kinase is one of the top drug targets for promoting health and lifespan extension, and AI-predicted mTOR inhibitors have already shown the ability to reduce cancer cell proliferation and extend lifespan in model organisms.
The PEARL trial, looking at rapamycin’s effects on visceral fat, bone mass density, and lean body mass in older adults, will provide the kind of rigorous human data that has been missing. The PEARL trial, a randomized, placebo-controlled phase 4 trial aiming to enrol 1,000 healthy older adults, will assess the safety and efficacy of rapamycin in improving visceral fat content, bone mass density, and lean body mass.
Genetic research, like the German longevity study, is giving researchers a map of which specific mTOR pathway components most influence human lifespan, allowing more targeted interventions rather than blunt pathway suppression.
The genetic findings could help researchers develop more targeted lifestyle recommendations that optimize mTOR signalling, and the most effective longevity interventions might involve targeting mTOR alongside other ageing-related pathways, creating synergistic effects that address multiple aspects of the ageing process simultaneously.
The Bottom Line on mTOR
Longevity researchers keep talking about mTOR because the evidence keeps pointing back to it. It sits at the centre of how cells decide to grow versus repair, how dietary signals translate into cellular behaviour, how ageing unfolds at the molecular level, and how we might one day slow that process in ways that matter clinically.
The honest summary is this: chronic mTOR overactivation, driven by caloric excess, sedentary behaviour, and constant protein availability, is probably one of the more significant accelerators of biological ageing in modern life. The evidence for that is strong. The evidence for any single pharmacological intervention to fix it in humans is still accumulating.
What is clear right now is that how you eat, when you eat, and how much you ask your cells to be in constant growth mode have measurable consequences for how you age. That is not marketing. It is the biological logic of one of the most studied molecular pathways in modern science, explained by a decade of research that has consistently pointed in the same direction.

